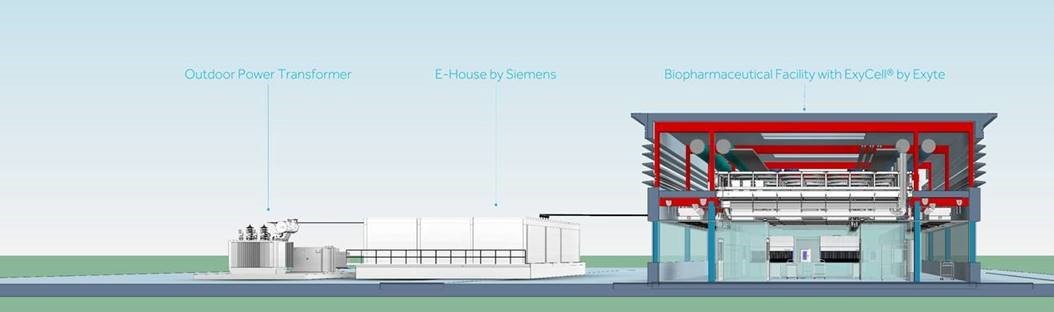
Against the background of the current challenges associated with the Covid-19 pandemic, pharmaceutical companies are facing challenges in developing and mass-producing new vaccines and medicines in existing production facilities. Siemens and Exyte are committed to tackle those challenges. Together, they offer standardized, turnkey solutions compliant with cGMP (current Good Manufacturing Practice) and GAMP (Good Automated Manufacturing Practice) to biotechnology manufacturers as well as cell and gene therapy manufacturers.
Modular technology for rapid construction
“We are excited to collaborate with Exyte to provide pre-fabricated, modular biotechnology solutions with our technology already embedded. As a market leader for the pharmaceutical industry, we offer know-how for process automation with our Simatic PCS 7 and WinCC automation platforms, as well as power supply and fire protection for modular cleanrooms,” said Eckard Eberle, CEO Siemens Process Automation.
Luca Mussati, Vice President Pharmaceuticals & Biotechnology at Exyte states: “Our partnership with Siemens enables us to pre-integrate Siemens’ technology into ExyCell modules, thus offering clients end-to-end solutions for their facilities. Our collaboration allows our clients to reap the benefits of industry 4.0 without the necessity of engineering them from scratch each time, thus saving them time and money. ExyCell modules are suitable for new buildings as well as for the retrofit of existing buildings, and can be provided either in standard, off-the-shelf plant configurations, or modules that can be combined to meet specific customer requirements.”
Currently, the first buildings with Siemens’ technology and ExyCell modules are being designed for cell and gene therapy manufacturing and biologicals production in China and Europe.